MEDIFUGE
MF 200 - MF 200 100
Medical Device
Centrifuge for the production of fibrin clots.
Class IIa medical device.
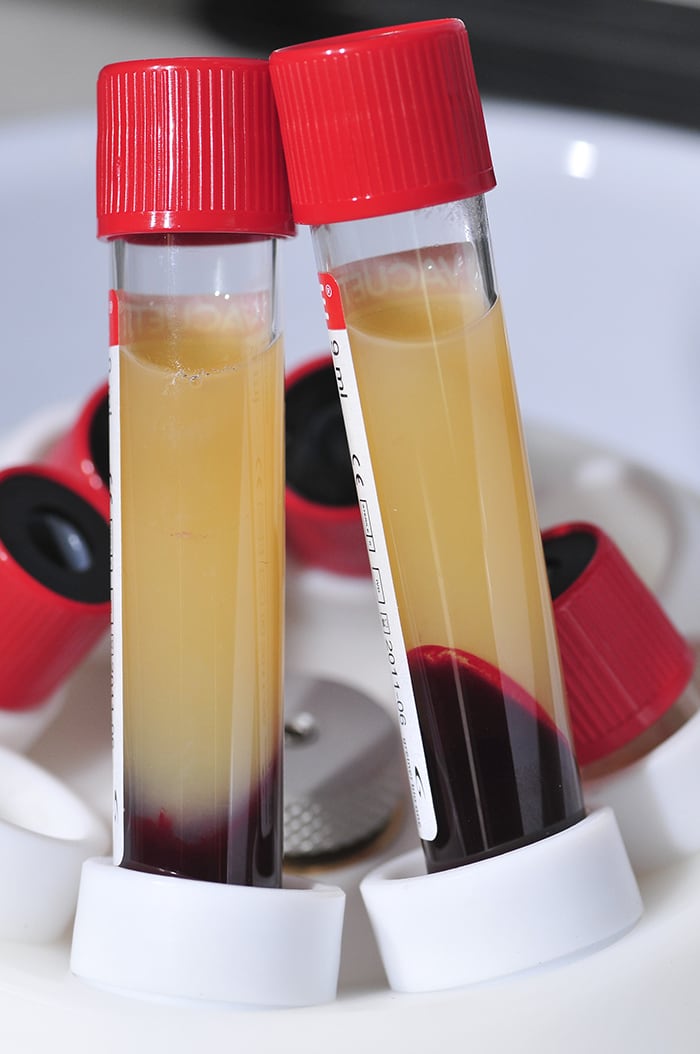
Blood Phase Separator .
The medical device MEDIFUGE allows for the use of up to 8 test tubes for the creation of CGF (fibrin).
- A microprocessor control system allows for the maintaining of a constant speed;
- The exception rotor system with self-ventilation protects the blood sample from heat exposure. The rotor-holding compartment, the lid and the test tube holders guarantee biological safety in terms of bio-containment, in the event of test tube breakage;
- The test tube holders and rotor are built from thermal, antistatic material that is easy to clean, extract and sterilise in an autoclave at 135°;
- MEDIFUGE is equipped with a decontamination cycle with UVC germicidal reflected light;
- Cycle duration 5 minutes at 1,000 revs;
- The electronic control engine and its internal parts require no maintenance;
- Noise levels fall below the standards required and do not exceed 57 dBa;
- Three predefined, non-editable programs: CGF-PRP1-PRP2
Technical features MF 200
- Power source: 230V 50/60 Hz;
- Nominal power consumption: 120 VA;
- Weight: 9,4 Kg;
- Dimensions: WxDxH: cm 32x23x24.
Technical features MF 200 100 (optional)
- Power source: 100-115V 50/ 60 Hz;
- Nominal power consumption: 120 VA;
- Weight: 9,4 Kg;
- Dimensions: WxDxH: cm 32x23x24.
Scope of use
- Parodontology
- Implantology
- Maxillo facial surgery
- Cosmetic surgery
- General surgery
- Wound care
- Ulcer care
CGF KIT
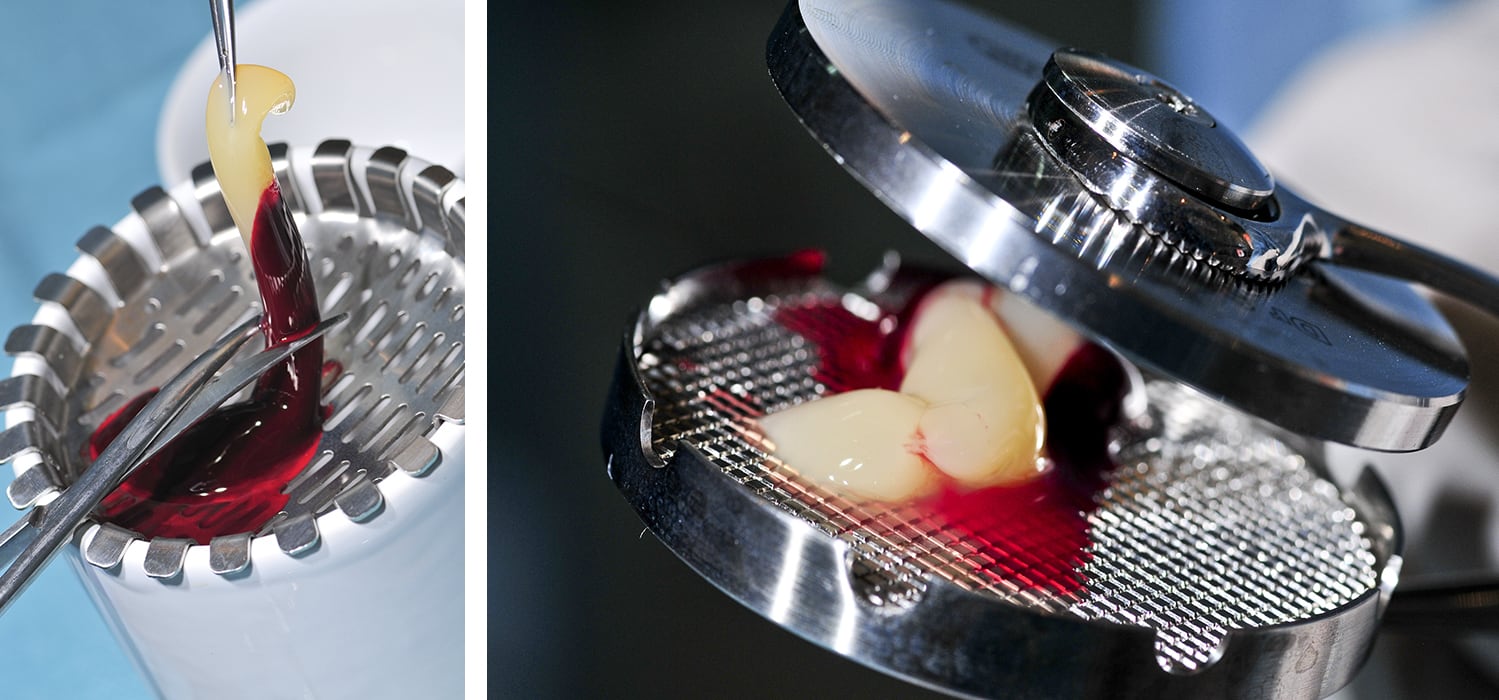
 CGF – Concentrated Growth Factors – represents a new generation of platelet holding inside a higher concentration of autologous growth factor. It is isolated from blood samples through a simple standardized separation protocol, which is performed by means of a specific centrifuge without the addition of exogenous substances.
CGF – Concentrated Growth Factors – represents a new generation of platelet holding inside a higher concentration of autologous growth factor. It is isolated from blood samples through a simple standardized separation protocol, which is performed by means of a specific centrifuge without the addition of exogenous substances.
The patent kit includes the necessary for the blood collection and specific dappens to facilitate the processing of hematic components without any contamination risks. Then, the kit is equipped with specific instruments to produce a fibrin membrane and to insert the clot inside the implant site.
CGF efficacy has been so far demostrated in oral and maxillofacial surgery, in maxillary sinus lift procedure and profile ridge augmentation.
Art. PV 200R
Art. CGF K1 LCE

Art. CGF K1 AGO
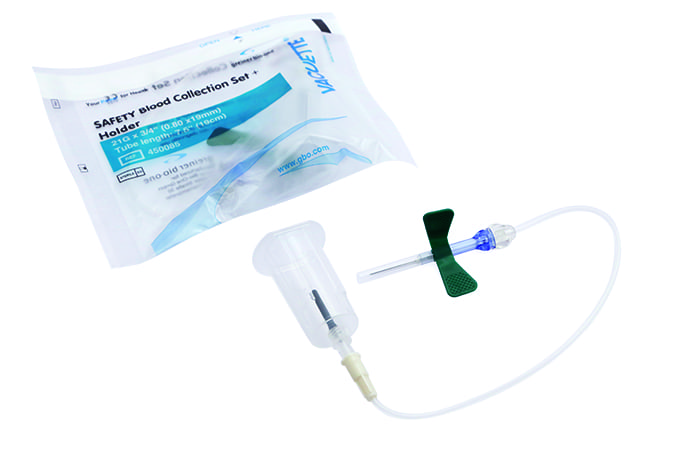
Art. CGF K1 CER01

Art. CGF K1 TMP02
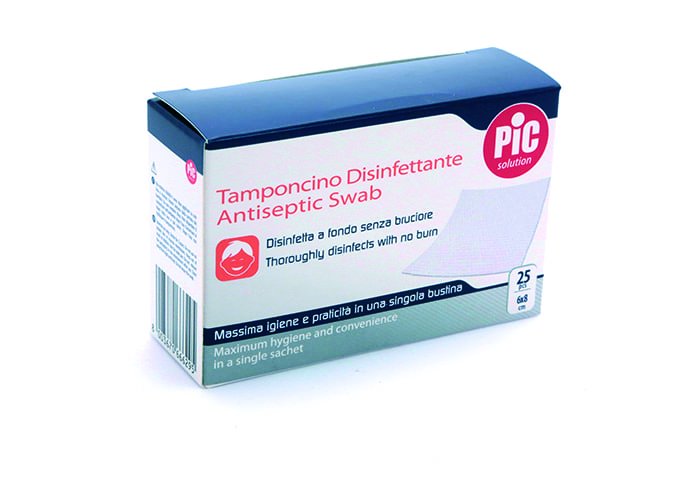
Art. CGF K1 PPR

Art. CGF K1 SPF

Art. CGF K1 GSP

Art. CGF K1 DFD

Art. CGF K1 DPC

Art. CGF K1 DPT

Art. CGF K1 FPS

Art. CGF K1 PAD

Art. CGF K1 SPD

Art. CGF K1 FMP

Art. CGF K1 COM

Art. CGF K1 PMS

Art. CGF K1 TRA

Related Products
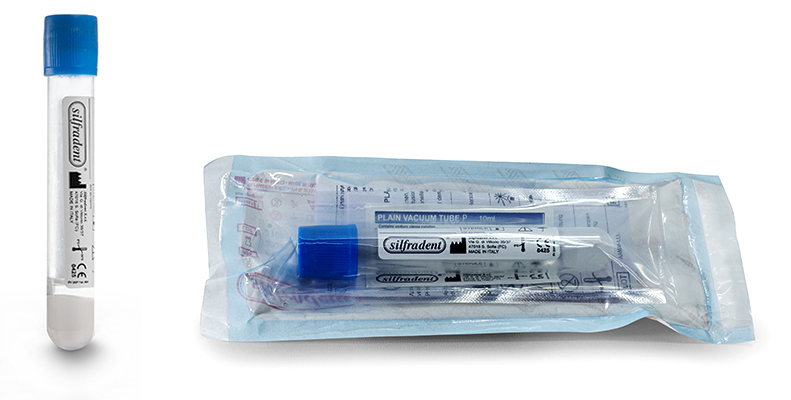
Test tube PV 200P (unitary code) 10 ml
Glass test tube with separator gel and sodium citrate anticoagulant: to produce liquid plasma PRP (Platelet Rich Plasma).
Pack of 20 test tubes: single unit packed in double sterile bag.
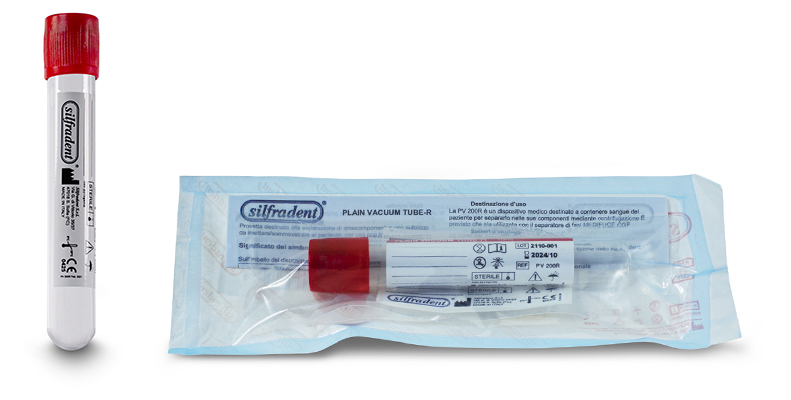
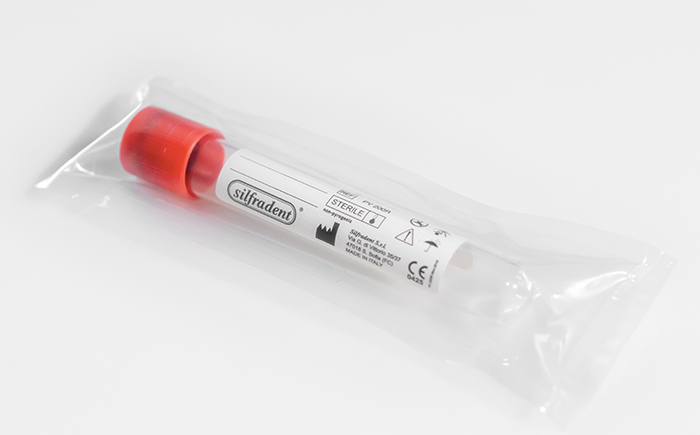
Test tube PV 200R (unitary code) 10 ml
Glass plain test tube empty to produce CGF (Concentrated Growth Factors) solid Plasma.
Pack of 50 test tubes: single unit packed in double sterile bag.